COVID-19
In September 2022, AnGes decides discontinuation of development of DNA vaccine for original Wuhan strain of COVID-19
and start of research into improved DNA vaccine for COVID-19 Variants and Formulation for its intranasal delivery.
Information on COVID-19
It was decided that we should utilize the plasmid DNA technology developed by our company as quickly as possible to respond the urgent global challenge of preventing the spread of COVID-19 and formulate effective solutions; accordingly, we announced "Joint Development of a DNA Vaccine to Combat the Covid-19" on March 5, 2020.
Relevant information, including the matters we have worked on since the announcement, and answers to the questions we have received are set out below.
[DNA vaccine for COVID-19]
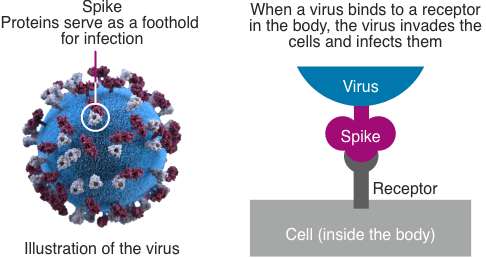
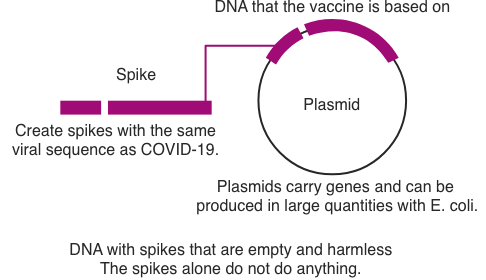
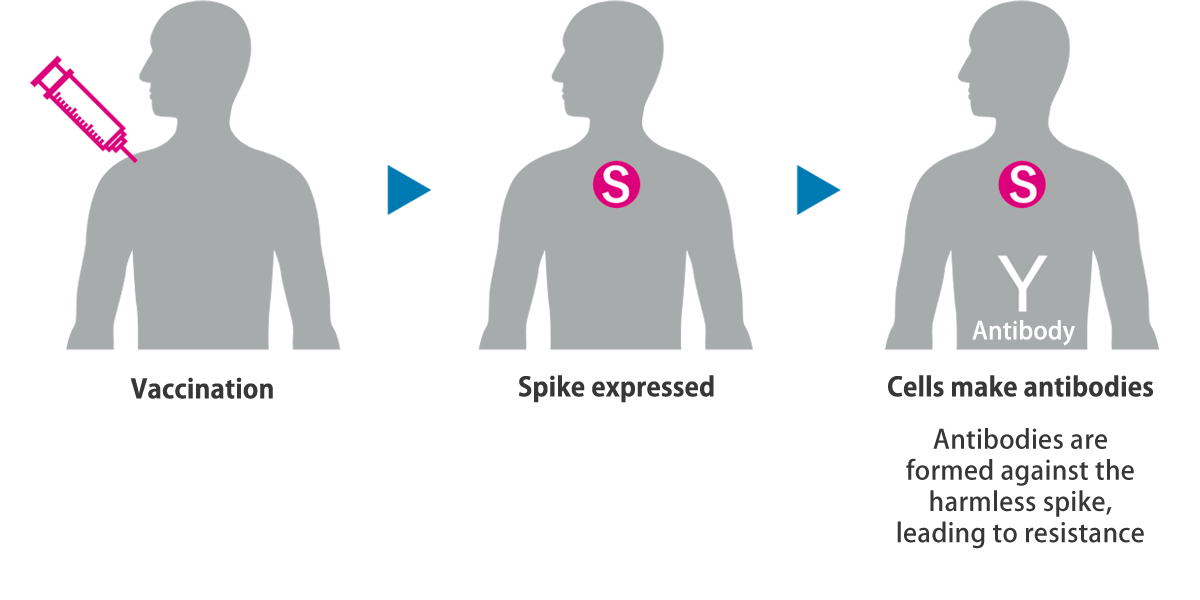
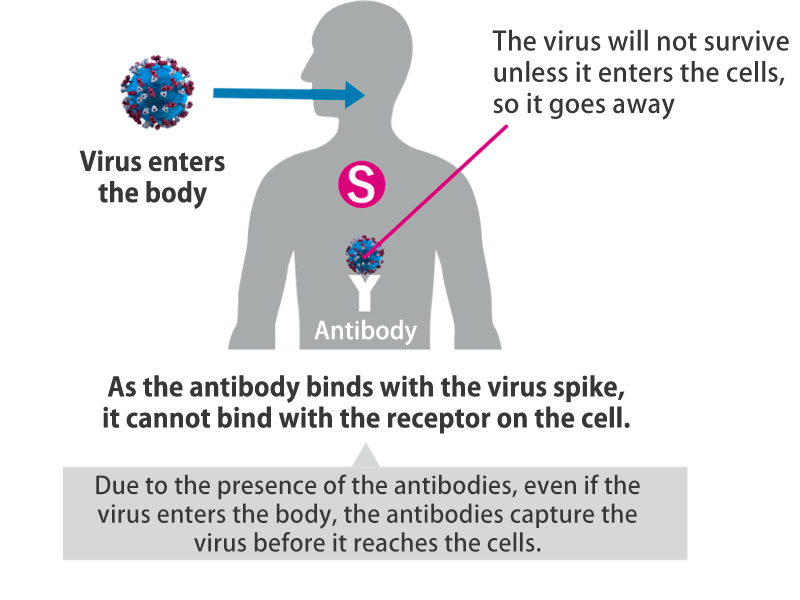
The DNA vaccine against COVID-19 is a plasmid DNA vaccine encoding the SARS-CoV-2 spike protein that opens the door to infection. It is expected to prevent COVID-19 or the condition of COVID-19 patients from deteriorating. Since the vaccine is engineered to express only the spikes on the viral surface, thereby producing antibodies, it is nonpathogenic and safe.
What are viruses?
What are DNA vaccines?
The vaccine produces antibodies by expressing only the spikes within the body.
COVID-19 News
- 2022.09.16IR
- About the September 7, 2022 Announcements
- 2021.11.05Development
- Novel Coronavirus (COVID-19) DNA Vaccine: Results of Phase 1/2 and Phase 2/3 Clinical Trials
- 2020.12.18Development
- Initiation of the First-in-Human Clinical Trial of a Potential Vascular Normalization COVID-19 Treatment
- 2020.11.13Development
- FDA Allowance of IND Application For AV-001 for the Treatment of Hospitalized Patients with COVID-19
- 2020.09.08Development
- COVID-19 DNA vaccine development_Announcement of Collaboration Agreement with Brickell Biotech
- 2020.04.14Development
- COVID-19 DNA vaccine project : Peptite Institute joins for the peptide synthesis research
- 2020.04.10Development
- COVID-19 DNA vaccine project : Listed on the COVID-19 candidate vaccines in WHO (World Health Organization)
- 2020.04.07Development
- COVID-19 DNA vaccine project: Participation of EPS Group to support the clinical trials
- 2020.03.24Development
- COVID-19 DNA vaccine project: Completion of Plasmid DNA vaccine for preclinical studies
